

January 11, 2021 from FDA:
Recall Limited to Lavva Blueberry Plant-Based Yogurt 5.3 Ounce Dated 2/21/21
Out of an abundance of caution, EVR Foods, INC, parent company to Lavva, is issuing a voluntary recall on its 5.3 ounce Blueberry Plant-Based Yogurt with expiration date 2/21/21. While this SKU and date code cleared strict quality assurance protocols, recent testing indicates a potential mold contamination. No other Lavva products are affected by the recall.
This plant-based yogurt was produced at a manufacturing facility in Norwich, NY, and has only been linked to the single date code. There have been no confirmed illnesses to date.
The recalled Blueberry Plant-Based Yogurts were distributed to retail stores nationwide. The product comes in a 5.3 ounce, colorful plastic cup with a foil lid, marked with lot #022121 on the bottom of the cup which is also used as the expiration date of the product.
Consumers are urged not to eat the product subject to the recall. Anyone who purchased the SKU may return it to the place of purchase for a full refund. Consumers with questions about the recall can reach out to hello@lovvelavva.com or 833-885-2882 between 9 a.m. to 5 p.m. EST Monday through Friday. The Food & Drug Administration (FDA) has been notified of this recall and is assisting with the process.


January 10, 2021 from FDA:
Weis Markets today said it has issued a recall for:
The Weis Quality Cookies and Cream Ice Cream (in 48 oz. containers) product has been removed from sale. It was sold in 197 Weis Markets’ stores in Pennsylvania, Maryland, Virginia, New Jersey, New York, Delaware and West Virginia.
The Weis Quality Cookies and Cream ice cream is packaged in a scround 48- ounce container with a UPC of 041497-01253 and with a sell by date of 10/28/21. This was produced on 10/28/2020 and released for sale on 10/29/2020. The sell by date is located on the bottom of the container. An example of the label has been included with this release. Customers who have purchased this product may return it for a full refund.
The Klein’s Vanilla bulk ice cream is packaged in 3-gallon containers with a code stamp of 0302 and are not for retail sale. This product was sold to one retail establishment in New York and has been removed from sale.
Additional ice cream products packaged on 10/28/2020 included:
These items are packaged in 3-gallon bulk containers with a code stamp of 0302 and are not for retail sale. They were stored in a warehouse and have not been distributed.
Customers requiring additional information may contact Weis Customer Service at 1-866-999-9347 Monday through Friday 8am-5pm EST.


November 25, 2020: From the Beth Din of Johannesburg:

January 9, 2021 from CFIA
Monaghan Mushrooms Ltd. is recalling Belle Grove brand Whole White Mushrooms from the marketplace because they may permit the growth of Clostridium botulinum. Consumers should not consume the recalled product described below.
| Brand | Product | Size | UPC | Codes |
|---|---|---|---|---|
| Belle Grove | Whole White Mushrooms | 227 g | 8 87462 00000 3 | Best Before 15 Jan |

December 30, 2020 - London Beth Din:


December 31, 2020 from the South African Beth Din:
Kosher With or Without a BD Logo:
December 29, 2020 from health.gov.il
Mizra Delicacies wishes to inform the consumers public that because of a suspicion of presence of a metal foreign body in the product, the product must not be consumed and should be returned to the points of sale.
This is a product that is usually sold in delis, not in its entirety, and is sliced in the delis.
Product name: Salami Kupecheski Servelat, institutional packaging weighing 1kg.
Production code: 20324
Best before: 19.03.2021
Product barcode: 7290004050368
Batch number written on the package: 2000012290
Anyone holding this product may return it to the point of sale and receive a different product in its place.The company is acting to recall the product from the marketing channels.For any question you can contact our customer serviceat *9872
December 28, 2020 from health.gov.il
Public announcement by Maadanei Galil Co.:
Selected Agricultural Produce Ltd. announces a recall of the product “Frozen Ground Chicken”.
An examination conducted by the Tel Aviv Health Bureau's food service foundbacteria of the type Salmonella in the product “Frozen Ground Chicken”, 500-gram package, produced on the 02.12.2020 (best before 01.02.2021)
Manufacturer: “Selected Agricultural Produce Ltd.” Barcode: 3415663. Customers in possession of the product are requested not to consume it. In coordination with the Food Service at the Ministry of Health, “Selected Agricultural Produce Ltd.” is performing a recall of the product from the marketing channels.
Customer service: 03-7357100

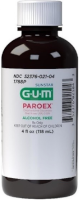
December 28, 2020 from FDA:
Schaumburg, Illinois, Sunstar Americas, Inc. (SAI) is voluntarily recalling Paroex® Chlorhexidine Gluconate Oral Rinse USP, 0.12% products bearing an expiration date from 12/31/2020 – 9/30/2022 to the consumer level. This product may be contaminated with the bacteria Burkholderia lata. This is an expansion of the recall initially announced on October 27, 2020.
To date, 29 adverse events have been reported to SAI related to this recall. Affected patients tested positive for Burkholderia lata infections, typically found in sputum cultures while under treatment for other serious medical conditions. Use of the contaminated product on patients with pre-existing respiratory conditions, including those infected with Covid-19, is particularly unsafe.
The prescription oral rinse product, available through healthcare professionals only, is indicated for use as part of a professional program for the treatment of gingivitis and is packaged as follows:
Consumers with questions regarding this recall can contact SAI by phone at 1-800-528-8537 or email us.pcr@us.sunstar.com on Monday-Friday from 8am-5pm CST. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to using this drug product.
Affected products and lot numbers follow below:
Product name:
Paroex® Chlorhexidine Gluconate Oral Rinse USP, 0.12%
Size/ Form:
16 fl.oz. Amber Bottles
NDC #:
052376-021-02
Product Code:
1789P
Lots Recalled:
ALL LOTS with expiration date from Dec. 31, 2020 through Sep. 30, 2022
| The information posted is from secondary sources. We cannot take responsibility for the accuracy of the information. |
Copyright © kashrut.com. Permission is granted to reprint these alerts in hardcopy print media if kashrut.com is credited as the source of the information.
כל האומר דבר בשם אומרו, מביא גאלה לעולם אבות ו"ו
| Comments to webmaster@kashrut.com
© Copyright 2024 Scharf Associates |
|
|||||||||||