
THIS SECTION IS FOR NEWS AND INTERESTING STORIES RELATED TO FOOD, NUTRITION AND FOOD PROCESSING. THEY ARE NOT NECESSARILY RELATED TO KOSHER BUT MAY BE OF INTEREST TO THE KOSHER CONSUMER, MANUFACTURER OR MASHGIACH.


May 20, 2021 from the FDA
This is to inform you of a product recall involving:
This recall has been initiated due to the presence of undeclared Methanol.
We began shipping this product on approximately July 10, 2020.
Immediately examine your inventory and quarantine product subject to recall. In addition, if you may have further distributed this product, please identify your customers and notify them at once of this product recall. Your notification to your customers may be enhanced by including a copy of this recall notification letter. If you have these recalled items in stock, please send an email to recall@globalsanitizers.com and we provide further instructions for product destruction.
Please email recall@globalsanitizers.com for further instructions. If you have any questions, call Ryan at 702.323.4402



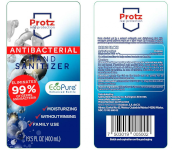
August 25, 2020 from the FDA
Mexico City, Mexico, Asiaticon SA de CV (Mexico) is voluntarily recalling all lots of V-Klean Hand Sanitizer Gel, Medically Minded Hand Sanitizer Gel and Protz Real Protection Antibacterial Hand Sanitizer sold in 13.5, 16.9 and 33.8 ounce bottles to the consumer level. The products are being recalled due to the potential presence of methanol (wood alcohol) and subpotent ethanol levels.
The product is used as a hand sanitizer and is packaged in plastic clear bottles with clear tops with UPC Code: V-Klean in 8.5 fl. oz. (250 ml) bottles: 716053704993 V-Klean in 16.9 fl. oz. (500 ml) bottles: 716053704993 V-Klean in 33.8 fl. oz. (1000 ml) bottles: 716053704993 Medically Minded in 16.9 fl. oz. (500 ml) bottles: 676753003782 Protz in 13.5 fl. oz. (400 ml) bottles : 7503019005002 The recall includes all lots. This product was exported to different distributors nationwide.
siaticon SA de CV is notifying its distributors by voluntary recall letter and consumers via this press release. Consumers that have the product subject to this recall should stop using and either contact Asiaticon SA de CV per the below for disposal instructions or return it to the place of purchase.
Consumers with questions regarding this recall can contact Asiaticon SA de CV at direccion@asiatic-connection.com (0052 1 55 21553488). In the US: at 929 394 3020 (available Monday to Friday 9.30 am 6 pm eastern time) Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to using this product.
FDA updates on hand sanitizers consumers should not use including list of recalled hand sanitizers.
| The information posted is from secondary sources. We cannot take responsibility for the accuracy of the information. |
| Comments to webmaster@kashrut.com
© Copyright 2026 Scharf Associates |
|
|||||||||||